InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the “Global Speciality & Orphan Drug Market Size, Share & Trends Analysis Report by Therapeutic Area (Oncology, Neurology & CNS Disorders, Hematology, Metabolic & Genetic Disorders, Lysosomal Storage Disorders, Immunology & Autoimmune Rare Diseases, Cardiovascular Rare Diseases, Infectious Diseases (Rare / Specialty), Rare Pediatric Diseases (Cross-segment), Respiratory Rare Diseases (CF, rare ILDs), Endocrine & Rare Hormonal Disorders), by Drug Modality (Small Molecules, Biologics, Cell & Gene Therapy, Nucleic Acid Therapies, Enzyme Replacement Therapy (ERT), Substrate Reduction Therapy, Peptides & Hormonal Therapies, Reformulated), by Dosage Form (Oral Solids (Tablets, Capsules), Oral Liquids / Suspensions /Solutions, Injectables, Long-Acting / Depot Formulations, Transdermal / Topical, Inhalation / Nasal, Advanced Delivery Systems), Distribution Channel (Speciality Pharmacy, Hospital / Buy-and-Bill, Retail Pharmacy (Limited), Hub Services (Patient Support Programs), Direct-to-Patient Distribution, Market Outlook And Industry Analysis 2035″
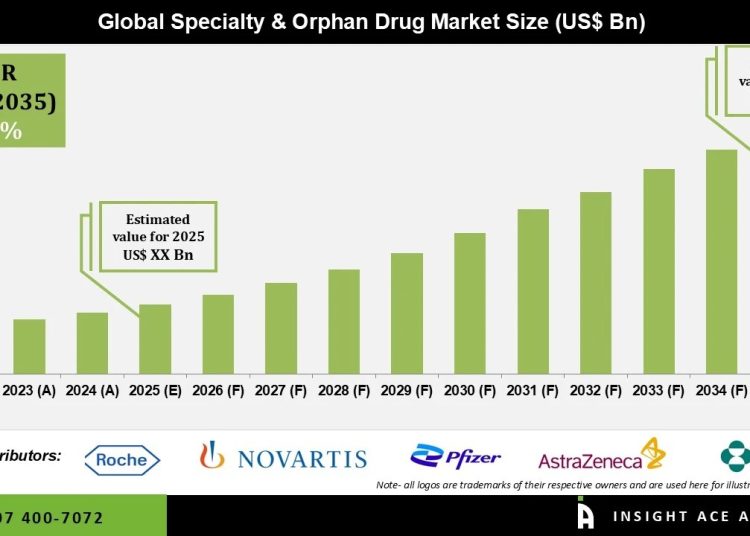
Speciality and Orphan Drug Market Size is predicted to reach grow at a 11.6% CAGR during the forecast period for 2026 to 2035.
Get Free Access to Demo Report, Excel Pivot and ToC: https://www.insightaceanalytic.com/request-sample/3519
The specialty and orphan drug market focuses on the development and commercialization of advanced therapies for rare, complex, and often life-threatening conditions affecting limited patient populations, including genetic disorders, rare malignancies, and autoimmune diseases. Pharmaceutical and biotechnology companies are increasingly prioritizing this segment, supported by favorable regulatory incentives such as orphan drug designation, expedited approval pathways, and extended market exclusivity. As a result, the market has evolved into a highly innovation-driven space with a strong emphasis on precision medicine and targeted therapeutic strategies.
In recent years, the sector has experienced significant advancement, driven by the introduction of biologics, gene therapies, and RNA-based treatments, which are transforming the management of rare diseases. Key industry players, including Novartis, Roche, and Vertex Pharmaceuticals, have been instrumental in advancing novel therapies, particularly for conditions such as spinal muscular atrophy, cystic fibrosis, and rare oncology indications.
These therapies often provide substantial clinical value, including the potential for long-term disease control or curative outcomes, thereby significantly improving patient quality of life. At the same time, advancements in genomics and diagnostic technologies have enhanced early detection and accurate diagnosis, enabling more effective and targeted treatment approaches.
Market expansion is further supported by rising demand for personalized medicine and increased awareness of rare diseases among both patients and healthcare providers. Collaborative efforts among pharmaceutical companies, academic institutions, and regulatory agencies have also contributed to shorter drug development timelines and improved patient access to innovative treatments.
Read Comprehensive Report Overview: https://www.insightaceanalytic.com/report/speciality-and-orphan-drug-market/3519
Despite these positive trends, the market continues to face challenges, including high treatment costs, limited patient populations, complex clinical trial designs, and reimbursement limitations. Nonetheless, the long-term outlook remains positive, driven by ongoing innovation, a robust development pipeline, and a growing global commitment to addressing unmet medical needs. Emerging developments such as gene-editing technologies, next-generation cell and gene therapies, and the integration of real-world evidence are expected to further reshape the landscape. These advancements are enhancing drug development efficiency and redefining treatment paradigms, positioning the specialty and orphan drug market as a vital component of modern healthcare and precision medicine.
List of Prominent Players in the Speciality & Orphan Drug Market:
- Roche
- Novartis
- Pfizer
- Johnson & Johnson
- AstraZeneca
- Merck & Co.
- Sanofi
- Takeda Pharmaceutical
- Bristol Myers Squibb
- Eli Lilly and Company
- Vertex Pharmaceuticals
- BioMarin Pharmaceutical
- Ultragenyx Pharmaceutical
- argenx
- Alnylam Pharmaceuticals
- Ionis Pharmaceuticals
- Sarepta Therapeutics
- Blueprint Medicines
- Incyte
- Azurity Pharmaceuticals
- Horizon Therapeutics
- Chiesi Farmaceutici
- Recordati Rare Diseases
- Amryt Pharma
- Supernus Pharmaceuticals
- Collegium Pharmaceutical
- Assertio Holding
Market Dynamics
Drivers:
The growth of the Specialty and Orphan Drug Market is strongly supported by advancements in genetic testing and diagnostic capabilities, which have enhanced the identification of rare diseases and expanded the potential patient base. Ongoing progress in biotechnology—particularly in gene editing, cell-based therapies, and RNA-based treatments—has enabled the development of highly targeted and effective therapeutic options.
In addition, the increasing involvement of biotechnology startups, coupled with strategic partnerships with established pharmaceutical companies, is accelerating both innovation and commercialization. The integration of digital technologies and advanced data analytics is improving clinical trial efficiency, while a growing focus on personalized and patient-centric healthcare is contributing to better treatment outcomes. Collectively, these factors are driving sustained expansion in the market.
Challenges:
Despite its strong growth trajectory, the Specialty and Orphan Drug Market faces several inherent challenges. Clinical trial execution is often complex due to the small and widely dispersed nature of patient populations, leading to higher recruitment costs and extended development timelines. Scientific uncertainties associated with rare diseases further add to the complexity of the drug development process.
Moreover, stringent regulatory requirements demand robust evidence of safety and efficacy, even in the context of limited clinical data. Advanced therapies, including gene- and cell-based treatments, require specialized infrastructure and controlled storage and distribution systems, increasing operational complexity. In certain regions, limited awareness and delayed diagnosis hinder timely access to treatment. Additionally, reliance on small patient populations raises concerns regarding long-term commercial sustainability, which can constrain market growth.
Regional Trends:
North America continues to hold a dominant position in the Specialty and Orphan Drug Market, supported by a well-established ecosystem characterized by advanced research capabilities, significant investment, and a highly developed healthcare infrastructure. The region benefits from comprehensive rare disease registries and real-world data platforms, which facilitate patient identification and streamline drug development processes.
A strong venture capital environment and active private investment further support early-stage biotechnology companies focused on innovative and high-risk therapies, fostering continuous advancement in the sector. In addition, a supportive regulatory framework with expedited review pathways enhances the efficiency and success rate of clinical development.
The widespread use of advanced diagnostic tools, including genetic testing, enables early and accurate identification of rare diseases, thereby increasing demand for targeted therapies. Furthermore, the presence of specialized treatment centers, integrated care networks, and well-developed cold-chain logistics infrastructure ensures the effective delivery and management of complex therapies. These combined factors reinforce North America’s leading position in the global specialty and orphan drug market.
Add our site to Google Preferred Sources for quality content: https://google.com/preferences/source?q=insightaceanalytic.com
Recent Developments:
- In September 2025, Novartis entered into multiple strategic collaborations in CAR-T therapy, radiotherapy, and RNA-based drugs to target rare cancers and genetic disorders.
- In July 2025, Vertex Pharmaceuticals expanded into gene-editing by advancing CRISPR-based therapies for rare diseases, enhancing its specialty drug pipeline.
Segmentation of Speciality & Orphan Drug Market-
By Therapeutic Areas –
- Oncology
- Rare Cancers
- Hematologic Malignancies
- Neurology & CNS Disorders
- SMA
- Epilepsy
- ALS
- Rare Neurogenetic
- Hematology
- Hemophilia
- Sickle Cell Disease
- Rare Anaemias
- Metabolic & Genetic Disorders
- Lysosomal Storage Disorders
- Pompe
- Gaucher
- Fabry
- Mitochondrial Diseases
- PKU & Urea Cycle Disorders
- Immunology & Autoimmune Rare Diseases
- Complement Disorders
- Primary Immunodeficiency
- Cardiovascular Rare Diseases
- ATTR Amyloidosis
- Pulmonary Arterial Hypertension
- Infectious Diseases (Rare / Speciality)
- Resistant infections
- HIV speciality segment
- Rare Pediatric Diseases (Cross-segment)
- Respiratory Rare Diseases (CF, rare ILDs)
- Endocrine & Rare Hormonal Disorders
By Drug Modality-
- Small Molecules
- Biologics
- Monoclonal Antibodies
- Bispecific Antibodies
- ADCs
- Cell & Gene Therapy
- CAR-T
- AAV Gene Therapy
- CRISPR
- Nucleic Acid Therapies
- ASO
- siRNA
- mRNA
- Enzyme Replacement Therapy (ERT)
- Substrate Reduction Therapy
- Peptides & Hormonal Therapies
- Reformulated
By Dosage Form –
- Oral Solids (Tablets, Capsules)
- Oral Liquids / Suspensions / Solutions
- Injectables
- IV
- Subcutaneous
- IM
- Long-Acting / Depot Formulations
- Transdermal / Topical
- Inhalation / Nasal
- Advanced Delivery Systems
- Implants
- Pumps
By Distribution Channel–
- Speciality Pharmacy
- Hospital / Buy-and-Bill
- Retail Pharmacy (Limited)
- Hub Services (Patient Support Programs)
- Direct-to-Patient Distribution
By Region-
North America-
Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Customize this Study according to your Requirements @ https://www.insightaceanalytic.com/customization/3519
About Us:
InsightAce Analytic is a market research and consulting firm that enables clients to make strategic decisions. Our qualitative and quantitative market intelligence solutions inform the need for market and competitive intelligence to expand businesses. We help clients gain competitive advantage by identifying untapped markets, exploring new and competing technologies, segmenting potential markets and repositioning products. Our expertise is in providing syndicated and custom market intelligence reports with an in-depth analysis with key market insights in a timely and cost-effective manner.
Contact us:
InsightAce Analytic Pvt. Ltd.
Visit: https://www.insightaceanalytic.com/
Tel : +1 607 400-7072
Asia: +91 79 72967118
info@insightaceanalytic.com